Q1. If all the peptide bonds of a protein are broken down, then what would remain?
Correct Answer: (a)
Peptide bonds link amino acids together to form proteins. Breaking all of them through hydrolysis results in individual free amino acids.
Peptide bonds link amino acids together to form proteins. Breaking all of them through hydrolysis results in individual free amino acids.
Q2. With regard to Carbohydrates, consider the given statements:
(1) They are classified on the basis of their behaviour on glycolysis.
(2) Polysaccharides are sweet sugars.
(3) All monosaccharides are reducing sugars
(1) They are classified on the basis of their behaviour on glycolysis.
(2) Polysaccharides are sweet sugars.
(3) All monosaccharides are reducing sugars
Correct Answer: (a)
Only statement (3) is correct. Monosaccharides are reducing sugars. Polysaccharides are not sweet. Classification is based on hydrolysis behavior, not glycolysis.
Only statement (3) is correct. Monosaccharides are reducing sugars. Polysaccharides are not sweet. Classification is based on hydrolysis behavior, not glycolysis.
Q3. Select false statements for an enzyme promoting a chemical reaction by
Correct Answer: (c)
Enzymes lower (not change) the activation energy and do not use heat or increased molecular motion (kinetic energy) to work; they work by stabilizing the transition state.
Enzymes lower (not change) the activation energy and do not use heat or increased molecular motion (kinetic energy) to work; they work by stabilizing the transition state.
Q4. In an experiment it was found that adenine constitutes 31% and guanine 19%. The quantity of cytosine in this DNA is likely to be
Correct Answer: (a)
According to Chargaff's rule in dsDNA, the amount of guanine (G) equals the amount of cytosine (C). If G is 19%, then C must be 19%.
According to Chargaff's rule in dsDNA, the amount of guanine (G) equals the amount of cytosine (C). If G is 19%, then C must be 19%.
Q5. Which one of the following is a non-reducing carbohydrate?
Correct Answer: (b)
Sucrose is a non-reducing sugar because the functional groups of both glucose and fructose are involved in the glycosidic linkage.
Sucrose is a non-reducing sugar because the functional groups of both glucose and fructose are involved in the glycosidic linkage.
Q6. Which of the following is wrongly matched ?
Correct Answer: (a)
Ribozymes are catalytic RNA molecules and are not proteinaceous in nature.
Ribozymes are catalytic RNA molecules and are not proteinaceous in nature.
Q7. Which one of the following vitamins serves as a coenzyme in transamination and decarboxylation of amino acid metabolism?
Correct Answer: (c)
Pyridoxine (Vitamin B6), in its active form pyridoxal phosphate, is the essential coenzyme for transamination and decarboxylation of amino acids.
Pyridoxine (Vitamin B6), in its active form pyridoxal phosphate, is the essential coenzyme for transamination and decarboxylation of amino acids.
Q8. The simplest amino acid is _________.
Correct Answer: (a)
Glycine is the simplest amino acid, where the R-group is just a hydrogen atom.
Glycine is the simplest amino acid, where the R-group is just a hydrogen atom.
Q9. With reference to double stranded DNA, The following ratio is always constant for all species
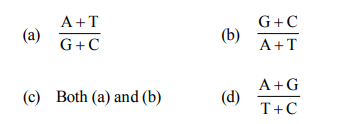
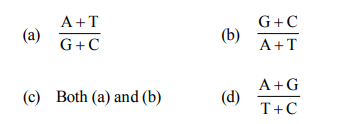
Correct Answer: (d)
According to Chargaff's rule, the ratio of Purines (A+G) to Pyrimidines (T+C) is always equal to one ($1:1$) in double-stranded DNA of all species.
According to Chargaff's rule, the ratio of Purines (A+G) to Pyrimidines (T+C) is always equal to one ($1:1$) in double-stranded DNA of all species.
Q10. The number of 'ends' in a glycogen molecule would be
Correct Answer: (a)
Because glycogen is highly branched, there is one non-reducing end for every branch, plus one reducing end for the whole molecule.
Because glycogen is highly branched, there is one non-reducing end for every branch, plus one reducing end for the whole molecule.
Q11. Enzymes are biocatalysts. They catalyse biochemical reactions. In general they reduce activation energy of reactions. Many physico-chemical processes are enzyme mediated. Some examples of enzyme mediated reactions are given below. Tick the wrong entry
Correct Answer: (a)
Dissolving CO2 in water is a physical process that occurs spontaneously, although the enzyme carbonic anhydrase can speed it up enormously in blood.
Dissolving CO2 in water is a physical process that occurs spontaneously, although the enzyme carbonic anhydrase can speed it up enormously in blood.
Q12. The diagram illustrates energy changes in an enzyme controlled reaction.
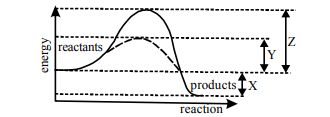
Which of the following represents the lowering of the activation energy?

Which of the following represents the lowering of the activation energy?
Correct Answer: (b)
Label Y represents the reduction in activation energy achieved by the enzyme (the difference between the two peaks).
Label Y represents the reduction in activation energy achieved by the enzyme (the difference between the two peaks).
Q13. Select the type of enzyme involved in the following reaction.
S — G + S' → S + S' — G
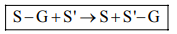
S — G + S' → S + S' — G
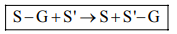
Correct Answer: (b)
The reaction shows the transfer of a functional group (G) from one substrate (S) to another (S'), which is the defining action of a transferase enzyme.
The reaction shows the transfer of a functional group (G) from one substrate (S) to another (S'), which is the defining action of a transferase enzyme.
Q14. A fat has
Correct Answer: (b)
A triglyceride (fat) is formed by the esterification of one glycerol molecule with three fatty acid molecules.
A triglyceride (fat) is formed by the esterification of one glycerol molecule with three fatty acid molecules.
Q15. Km is related to
Correct Answer: (b)
Km is a measure of the affinity of an enzyme for its substrate, describing the stability and formation of the Enzyme-Substrate (ES) complex.
Km is a measure of the affinity of an enzyme for its substrate, describing the stability and formation of the Enzyme-Substrate (ES) complex.
Q16. The steps in catalytic cycle of an enzyme action are given in random order.
(i) The enzyme releases the products. Now enzyme is free to bind another substrate.
(ii) The active sites, now in close proximity of substrate breaks the bond of substrate and forms E-P complex.
(iii) Binding of substrate induces the enzyme to alter its shape fitting more tightly around the substrate.
(iv) The substrate binds to the active site of enzyme (i.e., fitting into the active site).
(i) The enzyme releases the products. Now enzyme is free to bind another substrate.
(ii) The active sites, now in close proximity of substrate breaks the bond of substrate and forms E-P complex.
(iii) Binding of substrate induces the enzyme to alter its shape fitting more tightly around the substrate.
(iv) The substrate binds to the active site of enzyme (i.e., fitting into the active site).
Correct Answer: (b)
The correct sequence is: Substrate binds to active site (iv) → Induced fit (iii) → Catalysis/E-P formation (ii) → Product release (i).
The correct sequence is: Substrate binds to active site (iv) → Induced fit (iii) → Catalysis/E-P formation (ii) → Product release (i).
Q17. Amino acids, as the name suggests, have both an amino group and a carboxyl group in their structure. In addition, all naturally occurring aminoacids (those which are found in proteins) are called L-amino acids. From this, can you guess from which compound can the simplest amino acid be made?
Correct Answer: (b)
Amino acids are viewed as substituted methanes where four substituent groups occupy the four valency positions of the carbon atom.
Amino acids are viewed as substituted methanes where four substituent groups occupy the four valency positions of the carbon atom.
Q18. Consider the following statements:
(1) In proteins, only right handed helices are observed.
(2) In a polysaccharide chain, the right end is called the reducing end.
(3) B-DNA is generally coiled in left handed fashion.
Which of the statement(s) given above is/are incorrect?
(1) In proteins, only right handed helices are observed.
(2) In a polysaccharide chain, the right end is called the reducing end.
(3) B-DNA is generally coiled in left handed fashion.
Which of the statement(s) given above is/are incorrect?
Correct Answer: (a)
B-DNA is a right-handed helix. Left-handed DNA is known as Z-DNA. Statements (1) and (2) are correct biological facts.
B-DNA is a right-handed helix. Left-handed DNA is known as Z-DNA. Statements (1) and (2) are correct biological facts.
Q19. Ribozyme is a/an
Correct Answer: (b)
Ribozymes are catalytic RNA molecules that function as enzymes without being proteins.
Ribozymes are catalytic RNA molecules that function as enzymes without being proteins.
Q20. Phospholipids are important cell membrane constituents, because they
Correct Answer: (d)
Phospholipids are amphipathic molecules, meaning they have both a hydrophilic polar head and hydrophobic non-polar tails, allowing them to form stable bilayers in aqueous environments.
Phospholipids are amphipathic molecules, meaning they have both a hydrophilic polar head and hydrophobic non-polar tails, allowing them to form stable bilayers in aqueous environments.
Q21. Which of the following is an indispensable amino acid, and contains sulphur?
Correct Answer: (b)
Methionine is both an essential (indispensable) amino acid and one of the two sulfur-containing amino acids.
Methionine is both an essential (indispensable) amino acid and one of the two sulfur-containing amino acids.
Q22. Which one of the following biomolecules is correctly characterized?
Correct Answer: (a)
Lecithin is a phospholipid (phosphorylated glyceride) essential for cell membranes. Palmitic acid is saturated and has 16 carbons. Adenylic acid contains ribose, not glucose. In alpha-amino acids like alanine, the groups are attached to the same carbon.
Lecithin is a phospholipid (phosphorylated glyceride) essential for cell membranes. Palmitic acid is saturated and has 16 carbons. Adenylic acid contains ribose, not glucose. In alpha-amino acids like alanine, the groups are attached to the same carbon.
Q23. All the following amino acids are aromatic, except
Correct Answer: (d)
Tyrosine, phenylalanine, and tryptophan contain aromatic rings in their side chains. Valine is a branched-chain aliphatic amino acid.
Tyrosine, phenylalanine, and tryptophan contain aromatic rings in their side chains. Valine is a branched-chain aliphatic amino acid.
Q24. Match the protein given in column I with its function given in column II and choose the right option.
| Column I (Proteins) | Column II (Functions) |
|---|---|
| A. Collagen | I. Glucose transport |
| B. Trypsin | II. Hormone |
| C. Insulin | III. Intercellular ground substance |
| D. GLUT-4 | IV. Enzyme |
Correct Answer: (a)
Collagen is structural ground substance. Trypsin is a digestive enzyme. Insulin is a peptide hormone. GLUT-4 enables glucose transport into cells.
Collagen is structural ground substance. Trypsin is a digestive enzyme. Insulin is a peptide hormone. GLUT-4 enables glucose transport into cells.
Q25. When you take cells or tissue pieces and grind them with an acid in a mortar and pestle, all the small biomolecules dissolve in the acid. Proteins, polysaccharides and nucleic acids are insoluble in mineral acid and get precipitated. The acid soluble compounds include amino acids, nucleosides, small sugars etc. When one adds a phosphate group to a nucleoside one gets another acid soluble biomolecule called
Correct Answer: (d)
A nucleoside plus a phosphate group forms a nucleotide, which is found in the acid-soluble fraction of cellular extracts.
A nucleoside plus a phosphate group forms a nucleotide, which is found in the acid-soluble fraction of cellular extracts.
Q26. Enzymes are sensitive to
Correct Answer: (d)
Enzymes function optimally within narrow ranges of temperature and pH; significant deviations cause denaturation or loss of activity.
Enzymes function optimally within narrow ranges of temperature and pH; significant deviations cause denaturation or loss of activity.
Q27. Inulin is a polymer of
Correct Answer: (c)
Inulin is a storage polysaccharide found in several plants like dahlia, and it is a polymer of fructose.
Inulin is a storage polysaccharide found in several plants like dahlia, and it is a polymer of fructose.
Q28. Which one of the given graph shows the effect of pH on the velocity of a typical enzymatic reaction (V) ?
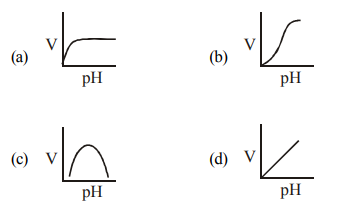

Correct Answer: (c)
Enzyme activity relative to pH or temperature typically follows a bell-shaped curve (c), with a peak at the optimum value and declining activity on either side.
Enzyme activity relative to pH or temperature typically follows a bell-shaped curve (c), with a peak at the optimum value and declining activity on either side.
Q29. Which one of the following pairs of nitrogenous bases of nucleic acids, is wrongly matched with the category mentioned against it ?
Correct Answer: (a)
Adenine and Guanine are Purines (double-ring). Thymine, Cytosine, and Uracil are Pyrimidines (single-ring).
Adenine and Guanine are Purines (double-ring). Thymine, Cytosine, and Uracil are Pyrimidines (single-ring).
Q30. Which of the following are not polymeric?
Correct Answer: (c)
Unlike proteins or DNA, lipids are not true polymers because they are not formed from repeating monomer chains, although they are macromolecules.
Unlike proteins or DNA, lipids are not true polymers because they are not formed from repeating monomer chains, although they are macromolecules.
Q31. Enzyme that break nucleic acids into nucleotides belongs to which of the following class ?
Correct Answer: (c)
Nucleases (which break nucleic acids) are hydrolases because they use water to cleave phosphodiester bonds.
Nucleases (which break nucleic acids) are hydrolases because they use water to cleave phosphodiester bonds.
Q32. Turn over number of enzyme depends upon
Correct Answer: (b)
Turnover number is the number of substrate molecules converted per unit time per active site of the enzyme.
Turnover number is the number of substrate molecules converted per unit time per active site of the enzyme.
Q33. The following diagrams represent the nitrogenous bases of nucleic acid molecules. Identify the correct combination:
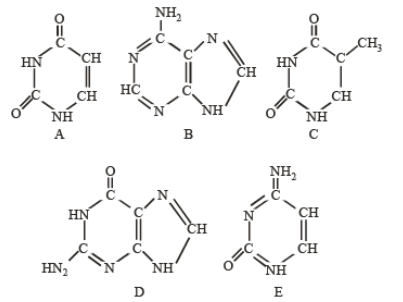

Correct Answer: (a)
A: Uracil, B: Adenine, C: Thymine, D: Guanine, E: Cytosine.
A: Uracil, B: Adenine, C: Thymine, D: Guanine, E: Cytosine.
Q34. Purines found both in DNA and RNA are:
Correct Answer: (b)
Adenine and Guanine are the two purines present in both DNA and RNA. Thymine and Uracil are pyrimidines unique to each.
Adenine and Guanine are the two purines present in both DNA and RNA. Thymine and Uracil are pyrimidines unique to each.
Q35. The figure given below show three velocity- substrate concentration curves for an enzyme reaction. What do the curves 1, 2 and 3 depict respectively?
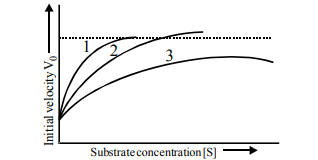

Correct Answer: (a)
Curve 1 is the control. Curve 2 shows Vmax is reached but requires more substrate (Competitive). Curve 3 shows a lower Vmax (Non-competitive).
Curve 1 is the control. Curve 2 shows Vmax is reached but requires more substrate (Competitive). Curve 3 shows a lower Vmax (Non-competitive).
Q36. Antiparallel strands of a DNA molecule means that
Correct Answer: (b)
Antiparallel means the two strands run in opposite directions; one 5' to 3' and the other 3' to 5', placing the starting phosphates at opposite ends.
Antiparallel means the two strands run in opposite directions; one 5' to 3' and the other 3' to 5', placing the starting phosphates at opposite ends.
Q37. Find out the wrongly matched pair
Correct Answer: (d)
Cellulose is a homopolymer composed strictly of glucose units linked by beta-1,4-glycosidic bonds.
Cellulose is a homopolymer composed strictly of glucose units linked by beta-1,4-glycosidic bonds.
Q38. Match column I (function) with column II (Types of enzymes) and select the correct option.
| Column I (Function) | Column II (Types of enzymes) |
|---|---|
| A. Enzymes catalysing breakdown without addition of water. | I. Isomerases |
| B. Enzyme catalyzes the conversion of an aldose sugar to a ketose sugar. | II. Oxidoreductase |
| C. Enzyme where catalysis involves transfer of electrons. | III. Ligases |
| D. Enzyme catalysing bonding of two components with the help of ATP. | IV. Lyases |
Correct Answer: (c)
Lyases break bonds without water. Isomerases catalyze structural rearrangements (aldose to ketose). Oxidoreductases involve electron transfer. Ligases join molecules using ATP energy.
Lyases break bonds without water. Isomerases catalyze structural rearrangements (aldose to ketose). Oxidoreductases involve electron transfer. Ligases join molecules using ATP energy.
Q39. Which one of the following structural formulae (A & B) of two organic compounds is correctly identified along with its related function?
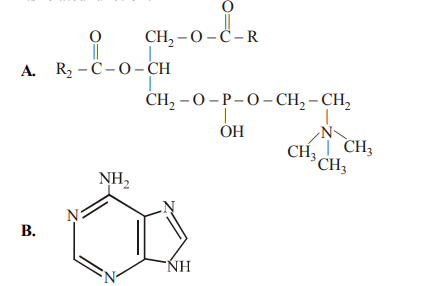

Correct Answer: (d)
Structure A is lecithin, a complex phospholipid that is a major structural component of cellular membranes. Structure B is adenine, which is a nitrogenous base, not a nucleotide itself.
Structure A is lecithin, a complex phospholipid that is a major structural component of cellular membranes. Structure B is adenine, which is a nitrogenous base, not a nucleotide itself.
Q40. Nucleotides are building blocks of nucleic acids. Each nucleotide is a composite molecule formed by
Correct Answer: (a)
A nucleotide is the monomeric unit consisting of a nitrogenous base, a pentose sugar, and a phosphate group.
A nucleotide is the monomeric unit consisting of a nitrogenous base, a pentose sugar, and a phosphate group.
Q41. The Km value of the enzyme is the value of the substrate concentration at which the reaction reaches to
Correct Answer: (c)
Repeating the definition of the Michaelis constant (Km) as the concentration where reaction velocity is half its maximum.
Repeating the definition of the Michaelis constant (Km) as the concentration where reaction velocity is half its maximum.
Q42. Match column I (category) with column II (secondary metabolites) and choose the correct option.
| Column I (Category) | Column II (Secondary metabolites) |
|---|---|
| A. Pigments | I. Concanavalin A |
| B. Terpenoides | II. Monoterpenes, Diterpenes |
| C. Alkaloids | III. Morphine, Codeine |
| D. Lectins | IV. Carotenoids, Anthocyanin |
Correct Answer: (a)
Carotenoids and anthocyanins are pigments. Monoterpenes and diterpenes are terpenoids. Morphine and codeine are alkaloids. Concanavalin A is a lectin.
Carotenoids and anthocyanins are pigments. Monoterpenes and diterpenes are terpenoids. Morphine and codeine are alkaloids. Concanavalin A is a lectin.
Q43. The quaternary structure of human haemoglobin is best described as a
Correct Answer: (d)
Human hemoglobin is a heterotetramer consisting of two pairs of different subunits (2 alpha and 2 beta chains).
Human hemoglobin is a heterotetramer consisting of two pairs of different subunits (2 alpha and 2 beta chains).
Q44. Identify the substances having glycosidic bond and peptide bond, respectively in their structure
Correct Answer: (c)
Inulin is a polysaccharide (glycosidic bonds). Insulin is a protein (peptide bonds).
Inulin is a polysaccharide (glycosidic bonds). Insulin is a protein (peptide bonds).
Q45. Consider the following statements:
(1) The structure of glycogen is similar to amylopectin.
(2) Unlike amylopectin, glycogen is soluble in water.
Which of the statement(s) given above is/are correct?
(1) The structure of glycogen is similar to amylopectin.
(2) Unlike amylopectin, glycogen is soluble in water.
Which of the statement(s) given above is/are correct?
Correct Answer: (a)
Glycogen and amylopectin are both branched polymers of glucose, though glycogen is more highly branched. Both are generally insoluble in water due to their large size.
Glycogen and amylopectin are both branched polymers of glucose, though glycogen is more highly branched. Both are generally insoluble in water due to their large size.
Q46. Inorganic catalyst work efficiently at _______temperature and ________ pressure.
Correct Answer: (d)
Unlike organic enzymes which denature at high heat, inorganic catalysts often require and operate efficiently at high temperatures and pressures.
Unlike organic enzymes which denature at high heat, inorganic catalysts often require and operate efficiently at high temperatures and pressures.
Q47. The charged molecule which is electrically neutral is known as
Correct Answer: (b)
A zwitterion is an ion that has separate positive and negative charges but a net charge of zero.
A zwitterion is an ion that has separate positive and negative charges but a net charge of zero.
Q48. Which of the following biomolecules have phosphodiester bond?
Correct Answer: (c)
Phosphodiester bonds link the 3' carbon of one sugar to the 5' carbon of the next in nucleic acid strands.
Phosphodiester bonds link the 3' carbon of one sugar to the 5' carbon of the next in nucleic acid strands.
Q49. Heart enlargement and paralysis can occur due to the deficiency of the following vitamin
Correct Answer: (b)
Deficiency of Vitamin B1 (Thiamine) leads to Beri-beri, which is characterized by neurological issues (paralysis) and cardiovascular complications (heart enlargement).
Deficiency of Vitamin B1 (Thiamine) leads to Beri-beri, which is characterized by neurological issues (paralysis) and cardiovascular complications (heart enlargement).
Q50. What will happen when the cofactor is removed from the enzyme?
Correct Answer: (a)
An apoenzyme (protein part) is catalytically inactive on its own; it requires its cofactor to form a functional holoenzyme.
An apoenzyme (protein part) is catalytically inactive on its own; it requires its cofactor to form a functional holoenzyme.
Q51. Concanavalin A is:
Correct Answer: (c)
Concanavalin A is a well-known lectin derived from the jack bean plant.
Concanavalin A is a well-known lectin derived from the jack bean plant.
Q52. The effectiveness of an enzyme is affected least by
Correct Answer: (c)
The enzyme's job is to lower the activation energy; the 'original' uncatalyzed energy level doesn't change the enzyme's efficiency itself, whereas temperature and concentrations directly affect reaction rates.
The enzyme's job is to lower the activation energy; the 'original' uncatalyzed energy level doesn't change the enzyme's efficiency itself, whereas temperature and concentrations directly affect reaction rates.
Q53. The given structural formulas represent amino acids (labelled by as X, Y and Z)
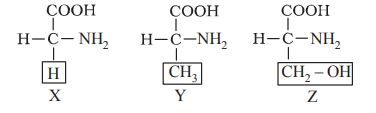
Identify the correct name of X, Y and Z.

Identify the correct name of X, Y and Z.
Correct Answer: (c)
In X, the R group is H (Glycine). In Y, the R group is CH3 (Alanine). In Z, the R group is CH2OH (Serine).
In X, the R group is H (Glycine). In Y, the R group is CH3 (Alanine). In Z, the R group is CH2OH (Serine).
Q54. Building block of nucleic acid is ________.
Correct Answer: (a)
Nucleic acids (DNA and RNA) are polymers made up of repeating units called nucleotides.
Nucleic acids (DNA and RNA) are polymers made up of repeating units called nucleotides.
Q55. Which of the following statement is wrong regarding chitin?
Correct Answer: (a)
Chitin is a structural polysaccharide, not a storage one. It provides rigidity to the fungal cell walls and the exoskeletons of arthropods.
Chitin is a structural polysaccharide, not a storage one. It provides rigidity to the fungal cell walls and the exoskeletons of arthropods.
Q56. The adjoining graph shows change in concentration of substrate on enzyme activity. Identify A, B and C.
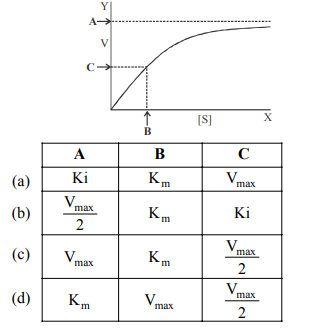

Correct Answer: (d)
In an enzyme saturation graph: A is the Maximum Velocity (Vmax), B is the substrate concentration at half Vmax (Km), and C is the point of Half Maximum Velocity (Vmax/2).
In an enzyme saturation graph: A is the Maximum Velocity (Vmax), B is the substrate concentration at half Vmax (Km), and C is the point of Half Maximum Velocity (Vmax/2).
Q57. The given graph shows concept of activation energy with labelled 1, 2, 3, & 4. Co-relate the statements I, II, III & IV with 1, 2, 3 & 4.
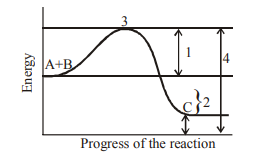

Correct Answer: (a)
1 represents Activation energy without enzyme. 2 represents Activation energy with enzyme. 3 is the Transition state. 4 represents the difference in energy between reactants and products (enthalpy change).
1 represents Activation energy without enzyme. 2 represents Activation energy with enzyme. 3 is the Transition state. 4 represents the difference in energy between reactants and products (enthalpy change).
Q58. Which of the following is not an aromatic amino acid?
Correct Answer: (a)
Threonine is a polar, uncharged amino acid. Tyrosine, tryptophan, and phenylalanine are the three aromatic amino acids.
Threonine is a polar, uncharged amino acid. Tyrosine, tryptophan, and phenylalanine are the three aromatic amino acids.
Q59. Which of the following glucose transporters is insulin dependent?
Correct Answer: (d)
GLUT IV is the insulin-regulated glucose transporter found primarily in adipose tissues and striated muscle.
GLUT IV is the insulin-regulated glucose transporter found primarily in adipose tissues and striated muscle.
Q60. Which of the following statement(s) is/are incorrect?
(i) Nearly 1/3rd of all enzymes requires the presence of metal ions for catalytic function.
(ii) Metal ions form coordinate bond with side chain at active site of metalloenzyme and at the same time form one or more coordinate bonds with the substrate.
(iii) NAD and NADP (coenzymes) contain niacin (vit.B3)
(iv) Coenzymes are organic compounds but their association with the apoenzyme is only transient, usually occurring during catalysis.
(i) Nearly 1/3rd of all enzymes requires the presence of metal ions for catalytic function.
(ii) Metal ions form coordinate bond with side chain at active site of metalloenzyme and at the same time form one or more coordinate bonds with the substrate.
(iii) NAD and NADP (coenzymes) contain niacin (vit.B3)
(iv) Coenzymes are organic compounds but their association with the apoenzyme is only transient, usually occurring during catalysis.
Correct Answer: (d)
All statements provided are correct biological descriptions of enzyme components and function according to standard textbooks.
All statements provided are correct biological descriptions of enzyme components and function according to standard textbooks.
Q61. Which of the following statements are correct?
(i) Chitin, a complex or heteropolysaccharide occuring in exoskeleton consists of NAG.
(ii) Glucosamine and N-acetyl glucosamine are modified sugar.
(iii) Cellulose shows blue colour when treated with I2.
(iv) Starch in plants and glycogen in animals are store houses of energy.
(v) Right end of polysaccharide is called reducing end while left end is called non-reducing end.
(i) Chitin, a complex or heteropolysaccharide occuring in exoskeleton consists of NAG.
(ii) Glucosamine and N-acetyl glucosamine are modified sugar.
(iii) Cellulose shows blue colour when treated with I2.
(iv) Starch in plants and glycogen in animals are store houses of energy.
(v) Right end of polysaccharide is called reducing end while left end is called non-reducing end.
Correct Answer: (c)
Statements (i), (ii), (iv), and (v) are correct. Cellulose does not show blue color with iodine because it lacks the helical structure necessary to hold iodine molecules; starch gives this reaction.
Statements (i), (ii), (iv), and (v) are correct. Cellulose does not show blue color with iodine because it lacks the helical structure necessary to hold iodine molecules; starch gives this reaction.
Q62. Which of the following is/are essential fatty acid(s)?
Correct Answer: (d)
Essential fatty acids like linoleic, linolenic, and arachidonic acids cannot be synthesized by the human body and must be obtained through the diet.
Essential fatty acids like linoleic, linolenic, and arachidonic acids cannot be synthesized by the human body and must be obtained through the diet.
Q63. Find out the correct match.
Correct Answer: (c)
Cellulose is the primary structural component of the plant cell wall. Inulin is a polymer of fructose. Glycogen is a polysaccharide and the storage form of energy in animals.
Cellulose is the primary structural component of the plant cell wall. Inulin is a polymer of fructose. Glycogen is a polysaccharide and the storage form of energy in animals.
Q64. Many organic substances are negatively charged e.g., acetic acid, while others are positively charged e.g., ammonium ion. An amino acid under certain conditions would have both positive and negative charges simultaneously in the same molecule. Such a form of amino acid is called
Correct Answer: (d)
The zwitterionic form is a specific state of an amino acid where it acts as a dipolar ion with equal numbers of positive and negative charges.
The zwitterionic form is a specific state of an amino acid where it acts as a dipolar ion with equal numbers of positive and negative charges.
Q65. Which of the following sugars have the same number of carbon as present in glucose?
Correct Answer: (a)
Both glucose and fructose are hexoses, meaning they both contain six carbon atoms (C6H12O6).
Both glucose and fructose are hexoses, meaning they both contain six carbon atoms (C6H12O6).
Q66. Transition state structure of the substrate formed during an enzymatic reaction is.
Correct Answer: (b)
The transition state is a high-energy, very brief (transient) intermediate that exists only during the reaction process and is inherently unstable.
The transition state is a high-energy, very brief (transient) intermediate that exists only during the reaction process and is inherently unstable.
Q67. Michaelis constant Km is equal to:
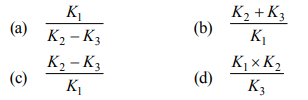
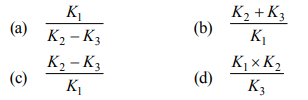
Correct Answer: (b)
The Km constant is derived from reaction rate constants as (K2 + K3) / K1, where K1 is the rate of ES formation, K2 is dissociation back to E+S, and K3 is product formation.
The Km constant is derived from reaction rate constants as (K2 + K3) / K1, where K1 is the rate of ES formation, K2 is dissociation back to E+S, and K3 is product formation.
Q68. Which of the following shows zwitter ionic form?
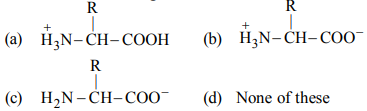
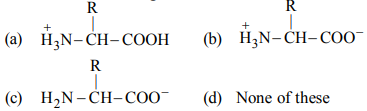
Correct Answer: (b)
A zwitterion is a dipolar ion where the amino group is protonated (NH3+) and the carboxyl group is deprotonated (COO-), resulting in an overall neutral charge.
A zwitterion is a dipolar ion where the amino group is protonated (NH3+) and the carboxyl group is deprotonated (COO-), resulting in an overall neutral charge.
Q69. Identify the basic amino acid from the following.
Correct Answer: (b)
Lysine, along with Arginine and Histidine, are classified as basic amino acids due to their side chain properties.
Lysine, along with Arginine and Histidine, are classified as basic amino acids due to their side chain properties.
Q70. Glycerol is a
Correct Answer: (b)
Glycerol is chemically known as trihydroxypropane, consisting of a three-carbon chain with a hydroxyl group (-OH) attached to each carbon.
Glycerol is chemically known as trihydroxypropane, consisting of a three-carbon chain with a hydroxyl group (-OH) attached to each carbon.
Q71. “All enzymes are proteins.” This statement is now modified because an apparent exception to this biological truth is
Correct Answer: (c)
The discovery of ribozymes (catalytic RNA) proved that not all biological catalysts are proteins.
The discovery of ribozymes (catalytic RNA) proved that not all biological catalysts are proteins.
Q72. Quaternary structure is present in ________.
Correct Answer: (a)
Adult hemoglobin is a quaternary protein consisting of four polypeptide subunits (two alpha and two beta chains).
Adult hemoglobin is a quaternary protein consisting of four polypeptide subunits (two alpha and two beta chains).
Q73. The two functional groups characteristic of sugars are
Correct Answer: (c)
Carbohydrates (sugars) are defined as polyhydroxy aldehydes or polyhydroxy ketones, containing multiple -OH and a C=O group.
Carbohydrates (sugars) are defined as polyhydroxy aldehydes or polyhydroxy ketones, containing multiple -OH and a C=O group.
Q74. A typical fat molecule is made up of
Correct Answer: (b)
A fat molecule (triglyceride) consists of one glycerol molecule esterified with three fatty acid chains.
A fat molecule (triglyceride) consists of one glycerol molecule esterified with three fatty acid chains.
Q75. Study the given statements and select the correct option.
(i) Nitrogen bases are open chain hydrocarbons.
(ii) A nucleoside differ from nucleotide in not having phosphate.
(iii) One turn of B-DNA has ten base pairs.
(iv) Length of one turn of DNA is 3.4 Å.
(i) Nitrogen bases are open chain hydrocarbons.
(ii) A nucleoside differ from nucleotide in not having phosphate.
(iii) One turn of B-DNA has ten base pairs.
(iv) Length of one turn of DNA is 3.4 Å.
Correct Answer: (d)
Nitrogenous bases are heterocyclic rings, not open chains. The length of one turn of B-DNA is 34 Å (3.4 nm), not 3.4 Å.
Nitrogenous bases are heterocyclic rings, not open chains. The length of one turn of B-DNA is 34 Å (3.4 nm), not 3.4 Å.
Q76. The bond between phosphate and hydroxyl group of sugar in nucleic acid is _______.
Correct Answer: (c)
A phosphate group reacts with the hydroxyl group of the sugar to form an ester bond. In a chain, these are called phosphodiester bonds.
A phosphate group reacts with the hydroxyl group of the sugar to form an ester bond. In a chain, these are called phosphodiester bonds.
Q77. Match the following:
(A) Inhibitor of catalytic activity - (i) Ricin
(B) Possess peptide bonds - (ii) Malonate
(C) Cell wall material in fungi - (iii) Chitin
(D) Secondary metabolite - (iv) Collagen
(A) Inhibitor of catalytic activity - (i) Ricin
(B) Possess peptide bonds - (ii) Malonate
(C) Cell wall material in fungi - (iii) Chitin
(D) Secondary metabolite - (iv) Collagen
Correct Answer: (d)
Malonate inhibits succinic dehydrogenase. Collagen has peptide bonds. Chitin is fungal wall material. Ricin is a secondary metabolite (toxin).
Malonate inhibits succinic dehydrogenase. Collagen has peptide bonds. Chitin is fungal wall material. Ricin is a secondary metabolite (toxin).
Q78. The enzyme hexokinase which catalysis glucose to a glucose-6-phosphate in glycolysis is inhibited by glucose 6-phosphate. This is an example of
Correct Answer: (c)
Product inhibition by glucose-6-phosphate on hexokinase is a classic example of feedback inhibition, where the product of a reaction regulates its own synthesis.
Product inhibition by glucose-6-phosphate on hexokinase is a classic example of feedback inhibition, where the product of a reaction regulates its own synthesis.
Q79. Which of the following statements is incorrect ?
Correct Answer: (b)
Lipids are generally water-insoluble (hydrophobic) and are instead soluble in organic solvents like ether or chloroform.
Lipids are generally water-insoluble (hydrophobic) and are instead soluble in organic solvents like ether or chloroform.
Q80. "When glucose is degraded to lactic acid in skeletal muscles energy is liberated". Which of the following conclusion is correctly associated with the above statement?
Correct Answer: (a)
The breakdown of glucose to lactic acid (glycolysis) is a catabolic pathway, which releases energy by breaking down complex molecules.
The breakdown of glucose to lactic acid (glycolysis) is a catabolic pathway, which releases energy by breaking down complex molecules.
Q81. Most abundant protein in human body is
Correct Answer: (a)
Collagen is the most abundant structural protein in the human body, found in skin, bone, and connective tissues.
Collagen is the most abundant structural protein in the human body, found in skin, bone, and connective tissues.
Q82. The catalytic efficiency of two different enzymes can be compared by the
Correct Answer: (a)
A lower Km indicates higher affinity for the substrate, which is a key measure of an enzyme's catalytic efficiency.
A lower Km indicates higher affinity for the substrate, which is a key measure of an enzyme's catalytic efficiency.
Q83. Which of the following set is correctly match to the category mentioned against them ?
Correct Answer: (d)
Cellulose, inulin, and glycogen are all complex carbohydrates (polysaccharides). Thiamine is a vitamin, not an amino acid. Acetic acid is not a fatty acid. Thymine is a base, not a nucleoside.
Cellulose, inulin, and glycogen are all complex carbohydrates (polysaccharides). Thiamine is a vitamin, not an amino acid. Acetic acid is not a fatty acid. Thymine is a base, not a nucleoside.
Q84. Feulgen reaction is based on which unique component of DNA
Correct Answer: (b)
The Feulgen reaction is a staining technique used to identify DNA; it specifically reacts with the aldehyde groups formed from the deoxyribose sugar after mild acid hydrolysis.
The Feulgen reaction is a staining technique used to identify DNA; it specifically reacts with the aldehyde groups formed from the deoxyribose sugar after mild acid hydrolysis.
Q85. A + G = C + T, then it is
Correct Answer: (d)
Chargaff's rule (Purines = Pyrimidines) specifically applies to double-stranded DNA (dsDNA).
Chargaff's rule (Purines = Pyrimidines) specifically applies to double-stranded DNA (dsDNA).
Q86. The pairing of A = T, and G ≡ C is the universal phenomenon of DNA. From this we can deduce all the followings except
Correct Answer: (a)
While =T$ and =C$ are true, the sum $(A+T)$ is not necessarily equal to $(G+C)$; this base composition ratio varies across different species.
While =T$ and =C$ are true, the sum $(A+T)$ is not necessarily equal to $(G+C)$; this base composition ratio varies across different species.
Q87. Which of the following statement is not true about amino acid?
Correct Answer: (c)
Amino acids are the components of peptides, but they themselves are not classified by the 'number of peptide bonds'. Instead, they are classified by the properties of their R-groups.
Amino acids are the components of peptides, but they themselves are not classified by the 'number of peptide bonds'. Instead, they are classified by the properties of their R-groups.
Q88. Which of the following vitamin is not having coenzyme activity?
Correct Answer: (d)
Vitamins D (Cholecalciferol), E (Tocopherol), and K (Menadione) are fat-soluble vitamins that generally act as hormones or antioxidants rather than specific metabolic coenzymes.
Vitamins D (Cholecalciferol), E (Tocopherol), and K (Menadione) are fat-soluble vitamins that generally act as hormones or antioxidants rather than specific metabolic coenzymes.
Q89. Which of the two groups (marked as 1, 2, 3, & 4) of following formula involved in peptide bond between different amino acids?
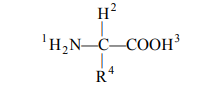
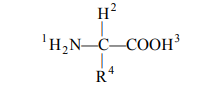
Correct Answer: (a)
A peptide bond forms between the amino group (-NH2, marked 2) of one amino acid and the carboxyl group (-COOH, marked 3) of the adjacent amino acid.
A peptide bond forms between the amino group (-NH2, marked 2) of one amino acid and the carboxyl group (-COOH, marked 3) of the adjacent amino acid.
Q90. Which of the following statements about enzymes is incorrect?
Correct Answer: (c)
While most enzymes are proteins, the non-protein exceptions are ribozymes (RNA), not lipids.
While most enzymes are proteins, the non-protein exceptions are ribozymes (RNA), not lipids.
Q91. The most abundant chemical in living organisms could be
Correct Answer: (b)
Water is the most abundant chemical substance in all living organisms, making up 70-90% of their total mass.
Water is the most abundant chemical substance in all living organisms, making up 70-90% of their total mass.
Q92. Which one of the following is wrongly matched?
Correct Answer: (c)
Enzymes are primarily proteins or RNA (ribozymes). Lipopolysaccharides are components of bacterial cell walls, not enzymes.
Enzymes are primarily proteins or RNA (ribozymes). Lipopolysaccharides are components of bacterial cell walls, not enzymes.
Q93. At temperature near freezing point, the enzymes are
Correct Answer: (a)
Low temperatures preserve enzymes in a temporarily inactive state, whereas high temperatures denature them.
Low temperatures preserve enzymes in a temporarily inactive state, whereas high temperatures denature them.
Q94. Unsaturated fatty acids have
Correct Answer: (d)
Unsaturated fatty acids are characterized by the presence of one or more double bonds between carbon atoms in their hydrocarbon chain.
Unsaturated fatty acids are characterized by the presence of one or more double bonds between carbon atoms in their hydrocarbon chain.
Q95. The acid used for preliminary separation of biomolecule in a living tissue is
Correct Answer: (c)
Chemical analysis of living tissue involves grinding it in trichloroacetic acid (Cl3CCOOH) to obtain a slurry, which is then filtered to separate acid-soluble and acid-insoluble fractions.
Chemical analysis of living tissue involves grinding it in trichloroacetic acid (Cl3CCOOH) to obtain a slurry, which is then filtered to separate acid-soluble and acid-insoluble fractions.
Q96. Feedback inhibition of enzymes is affected by
Correct Answer: (c)
Feedback inhibition occurs when the final product of a metabolic pathway inhibits an enzyme early in the pathway to regulate production.
Feedback inhibition occurs when the final product of a metabolic pathway inhibits an enzyme early in the pathway to regulate production.
Q97. Which of the following statements is correct?
Correct Answer: (d)
In DNA, Adenine (A) always pairs with Thymine (T) via two hydrogen bonds, while Guanine (G) pairs with Cytosine (C) via three.
In DNA, Adenine (A) always pairs with Thymine (T) via two hydrogen bonds, while Guanine (G) pairs with Cytosine (C) via three.
Q98. Biomolecules are
Correct Answer: (a)
By definition, biomolecules are the organic compounds found in living organisms that are essential for biological processes.
By definition, biomolecules are the organic compounds found in living organisms that are essential for biological processes.
Q99. According to weight percentage, the first three elements in human body are
Correct Answer: (d)
The human body is primarily composed of Oxygen (~65%), followed by Carbon (~18.5%) and Hydrogen (~9.5%) by weight.
The human body is primarily composed of Oxygen (~65%), followed by Carbon (~18.5%) and Hydrogen (~9.5%) by weight.
Q100. Glycogen is a homopolymer made up of
Correct Answer: (a)
Glycogen is strictly composed of repeating glucose monomers linked by glycosidic bonds.
Glycogen is strictly composed of repeating glucose monomers linked by glycosidic bonds.
Q101. Sugar-phosphate backbone in nucleic acid
Correct Answer: (d)
The sugar-phosphate backbone is highly polar and hydrophilic, carries negative charges due to phosphate groups, and has directionality (5' to 3' polarity).
The sugar-phosphate backbone is highly polar and hydrophilic, carries negative charges due to phosphate groups, and has directionality (5' to 3' polarity).
Q102. Match column I (organic compound) with column II (examples) and choose the correct combination from the given options.
| Column-I (Organic Compounds) | Column-II (Examples) |
|---|---|
| A. Fatty acid | I. Glutamic acid |
| B. Phospholipid | II. Tryptophan |
| C. Aromatic amino acid | III. Lecithin |
| D. Acidic amino acid | IV. Palmitic acid |
Correct Answer: (b)
Palmitic acid is a fatty acid. Lecithin is a phospholipid. Tryptophan is an aromatic amino acid. Glutamic acid is an acidic amino acid.
Palmitic acid is a fatty acid. Lecithin is a phospholipid. Tryptophan is an aromatic amino acid. Glutamic acid is an acidic amino acid.
Q103. Which of the given option is correct for the following statements?
(i) The metabolic pathway in which acetic acid is converted into cholesterol is an endothermic one.
(ii) Anabolic pathway is endergonic while catabolic pathway is exergonic.
(iii) Without metabolism there can not be a living state.
(i) The metabolic pathway in which acetic acid is converted into cholesterol is an endothermic one.
(ii) Anabolic pathway is endergonic while catabolic pathway is exergonic.
(iii) Without metabolism there can not be a living state.
Correct Answer: (a)
Metabolism is a defining feature of life. Anabolic processes (building) consume energy (endergonic), while catabolic processes (breaking) release energy (exergonic). Cholesterol synthesis is an anabolic, energy-consuming process.
Metabolism is a defining feature of life. Anabolic processes (building) consume energy (endergonic), while catabolic processes (breaking) release energy (exergonic). Cholesterol synthesis is an anabolic, energy-consuming process.
Q104. The primary structure of a protein molecule has
Correct Answer: (a)
Every linear protein chain has two distinct ends: the N-terminal (amino) end and the C-terminal (carboxyl) end.
Every linear protein chain has two distinct ends: the N-terminal (amino) end and the C-terminal (carboxyl) end.
Q105. The most basic amino acid is
Correct Answer: (a)
Arginine is the most basic amino acid due to its guanidino group in the side chain, which is highly alkaline.
Arginine is the most basic amino acid due to its guanidino group in the side chain, which is highly alkaline.
Q106. The curve given below shows enzymatic activity with relation to three conditions (pH, temperature and substrate concentration.) Identify the correct representation of two axes (x and y).
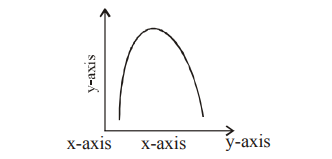

Correct Answer: (b)
The bell-shaped curve represents the effect of pH or temperature (on the x-axis) on the rate of enzyme reaction/velocity (on the y-axis).
The bell-shaped curve represents the effect of pH or temperature (on the x-axis) on the rate of enzyme reaction/velocity (on the y-axis).
Q107. The following diagrams represent the nitrogenous bases of nucleic acid molecules. Identify the correct combination
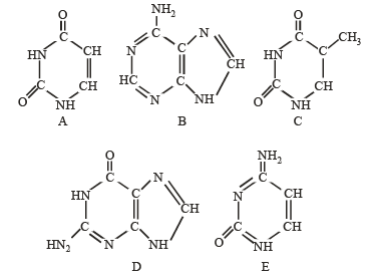

Correct Answer: (a)
A is Uracil (pyrimidine), B is Adenine (purine), C is Thymine (pyrimidine with methyl), D is Guanine (purine with carbonyl), and E is Cytosine (pyrimidine).
A is Uracil (pyrimidine), B is Adenine (purine), C is Thymine (pyrimidine with methyl), D is Guanine (purine with carbonyl), and E is Cytosine (pyrimidine).
Q108. A homopolymer has only one type of building block called monomer repeated 'n' number of times. A heteropolymer has more than one type of monomer. Proteins are heteropolymer usually made of
Correct Answer: (a)
Proteins are heteropolymers composed of various sequences of the 20 standard types of amino acids.
Proteins are heteropolymers composed of various sequences of the 20 standard types of amino acids.
Q109. Nucleoside is :
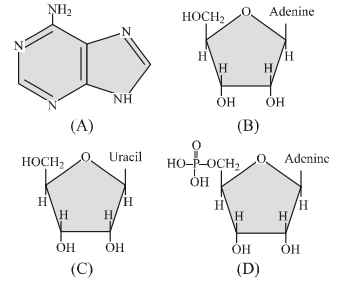

Correct Answer: (b)
In the diagrams: A is a nitrogenous base only, B and C are base + sugar (nucleosides), and D is base + sugar + phosphate (nucleotide).
In the diagrams: A is a nitrogenous base only, B and C are base + sugar (nucleosides), and D is base + sugar + phosphate (nucleotide).
Q110. Match column I with column II and choose the correct option.
| Column-I | Column-II |
|---|---|
| A. Cotton fibre | I. Chitin |
| B. Exoskeleton of cockroach | II. Glycogen |
| C. Liver | III. Starch |
| D. Peeled potato | IV. Cellulose |
Correct Answer: (b)
Cotton fiber is nearly pure cellulose. The exoskeleton of insects like cockroaches is made of chitin. Glycogen is stored in the liver. Potatoes are rich in starch.
Cotton fiber is nearly pure cellulose. The exoskeleton of insects like cockroaches is made of chitin. Glycogen is stored in the liver. Potatoes are rich in starch.
Q111. Which of the following statement is incorrect regarding secondary metabolites ?
Correct Answer: (c)
Abrin and ricin are classified as toxins, not drugs. Common drugs derived from secondary metabolites include vinblastine and curcumin.
Abrin and ricin are classified as toxins, not drugs. Common drugs derived from secondary metabolites include vinblastine and curcumin.
Q112. Which of the following sets contains polysaccharides?
Correct Answer: (b)
Starch, glycogen, and cellulose are all large polymers of glucose (polysaccharides). Glucose, fructose, and galactose are monosaccharides, while lactose, sucrose, and maltose are disaccharides.
Starch, glycogen, and cellulose are all large polymers of glucose (polysaccharides). Glucose, fructose, and galactose are monosaccharides, while lactose, sucrose, and maltose are disaccharides.
Q113. Select the incorrect statement.
Correct Answer: (c)
Lysine is a basic amino acid, not neutral, as it contains an additional amino group in its side chain.
Lysine is a basic amino acid, not neutral, as it contains an additional amino group in its side chain.
Q114. Which of the following is the example of acidic amino acid ?
Correct Answer: (d)
Glutamic acid and aspartic acid are both acidic amino acids because their R-groups contain an additional carboxyl group.
Glutamic acid and aspartic acid are both acidic amino acids because their R-groups contain an additional carboxyl group.
Q115. The amino acid Tryptophan is the precursor for the synthesis of
Correct Answer: (a)
Tryptophan is the metabolic precursor for the neurotransmitter serotonin and the hormone melatonin.
Tryptophan is the metabolic precursor for the neurotransmitter serotonin and the hormone melatonin.
Q116. Relation between amino acid and protein is similar to the one found between
Correct Answer: (d)
Just as amino acids are the monomeric units that build proteins, nucleotides are the monomers that build nucleic acids.
Just as amino acids are the monomeric units that build proteins, nucleotides are the monomers that build nucleic acids.
Q117. Which one of the following statements is incorrect?
Correct Answer: (c)
A competitive inhibitor increases the apparent Km of the enzyme because more substrate is needed to reach half Vmax; it does not decrease it.
A competitive inhibitor increases the apparent Km of the enzyme because more substrate is needed to reach half Vmax; it does not decrease it.
Q118. The given graph shows the effect of substrate concentration on the rate of reaction of the enzyme green -gram -phosphatase. What does the graph indicate ?
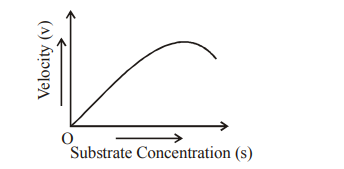

Correct Answer: (b)
The graph shows a typical sigmoidal or inhibited curve where the reaction rate is significantly lower than normal, indicating the presence of an inhibitor.
The graph shows a typical sigmoidal or inhibited curve where the reaction rate is significantly lower than normal, indicating the presence of an inhibitor.
Q119. A nucleotide is formed of
Correct Answer: (c)
A nucleotide is always composed of three parts: a nitrogenous base (either a purine or pyrimidine), a pentose sugar, and a phosphate group.
A nucleotide is always composed of three parts: a nitrogenous base (either a purine or pyrimidine), a pentose sugar, and a phosphate group.
Q120. On hydrolysis, a nucleoside would not yield
Correct Answer: (d)
A nucleoside contains only a nitrogenous base and a sugar. Phosphoric acid is only found in nucleotides.
A nucleoside contains only a nitrogenous base and a sugar. Phosphoric acid is only found in nucleotides.
Q121. Refer the given structure of adenylic acid. In this identify A.
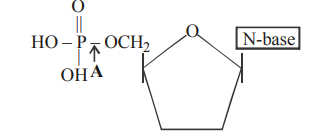

Correct Answer: (c)
The bond between the sugar's 5' hydroxyl group and the phosphate group in a nucleotide is a phosphoester bond (or simply an ester bond).
The bond between the sugar's 5' hydroxyl group and the phosphate group in a nucleotide is a phosphoester bond (or simply an ester bond).
Q122. A nucleoside differs from nucleotide is not having
Correct Answer: (c)
A nucleoside consists of a nitrogenous base and a pentose sugar. A nucleotide is a nucleoside with an added phosphate group.
A nucleoside consists of a nitrogenous base and a pentose sugar. A nucleotide is a nucleoside with an added phosphate group.
Q123. Primary structure of polypeptide is stabilized or secondary structure of polypeptide is maintained by
Correct Answer: (c)
Secondary structures like alpha-helices and beta-pleated sheets are primarily stabilized by hydrogen bonds between the peptide backbone components.
Secondary structures like alpha-helices and beta-pleated sheets are primarily stabilized by hydrogen bonds between the peptide backbone components.
Q124. Consider the following statements about Sucrose:
(1) Sucrose is laevorotatory but after hydrolysis the solution becomes dextrorotatory.
(2) The laevorotation of glucose is more than dextrorotation of fructose.
Which of the statement(s) given above is/are correct?
(1) Sucrose is laevorotatory but after hydrolysis the solution becomes dextrorotatory.
(2) The laevorotation of glucose is more than dextrorotation of fructose.
Which of the statement(s) given above is/are correct?
Correct Answer: (d)
Sucrose is dextrorotatory ($+66.5^\circ$) and becomes laevorotatory upon hydrolysis (inversion) due to the strong laevorotation of fructose ($-92.4^\circ$). Both statements provided are false.
Sucrose is dextrorotatory ($+66.5^\circ$) and becomes laevorotatory upon hydrolysis (inversion) due to the strong laevorotation of fructose ($-92.4^\circ$). Both statements provided are false.
Q125. Match the biomolecules given in column I with their examples given in column II and choose the correct answer.
| Column I (Biomolecules) | Column II (Examples) |
|---|---|
| A. Carbohydrates | I. Trypsin |
| B. Protein | II. Cholesterol |
| C. Nucleic acid | III. Inulin |
| D. Lipid | IV. Adenylic acid |
Correct Answer: (a)
Inulin is a carbohydrate (polysaccharide). Trypsin is a protein (enzyme). Adenylic acid is a nucleic acid (nucleotide). Cholesterol is a lipid (sterol).
Inulin is a carbohydrate (polysaccharide). Trypsin is a protein (enzyme). Adenylic acid is a nucleic acid (nucleotide). Cholesterol is a lipid (sterol).
Q126. Pick out the correct statement.
Correct Answer: (c)
Proteins are indeed linear polymers of amino acids linked by peptide bonds. RuBisCO is the most abundant protein in the biosphere (collagen is most abundant in animals). Chitin is a homopolymer of NAG. Polysaccharides use glycosidic bonds.
Proteins are indeed linear polymers of amino acids linked by peptide bonds. RuBisCO is the most abundant protein in the biosphere (collagen is most abundant in animals). Chitin is a homopolymer of NAG. Polysaccharides use glycosidic bonds.
Q127. Select the incorrect statement.
Correct Answer: (b)
Proteins are heteropolymers because they are composed of 20 different types of amino acids, not just one repeating type.
Proteins are heteropolymers because they are composed of 20 different types of amino acids, not just one repeating type.
Q128. It is said that elemental composition of living organisms and that of inanimate objects (like earth's crust) are similar in the sense that all the major elements are present in both. Then what would be the difference between these two groups?
Correct Answer: (c)
A relative abundance table shows that life selectively concentrates Carbon, Hydrogen, and Oxygen far more than the Earth's crust.
A relative abundance table shows that life selectively concentrates Carbon, Hydrogen, and Oxygen far more than the Earth's crust.
Q129. Proteins perform many physiological functions. For example some proteins function as enzymes. One of the following represents an additional function that some protein discharge
Correct Answer: (d)
Many vital hormones, such as insulin and growth hormone, are chemically proteins or polypeptides.
Many vital hormones, such as insulin and growth hormone, are chemically proteins or polypeptides.
Q130. Primary structure of protein is due to the presence of ______.
Correct Answer: (b)
The primary structure of a protein refers to the linear sequence of amino acids held together by covalent peptide bonds.
The primary structure of a protein refers to the linear sequence of amino acids held together by covalent peptide bonds.
Q131. Hexokinase (Glucose + ATP → Glucose 6-P + ADP) belongs to the category
Correct Answer: (a)
Transferases catalyze the transfer of a group (in this case, a phosphate group) from one substrate to another.
Transferases catalyze the transfer of a group (in this case, a phosphate group) from one substrate to another.
Q132. Which one of the following statements is wrong?
Correct Answer: (d)
Glycine does not contain sulfur; it is the simplest amino acid. Sulfur-containing amino acids are methionine and cysteine.
Glycine does not contain sulfur; it is the simplest amino acid. Sulfur-containing amino acids are methionine and cysteine.
Q133. Mark the odd pair in the followings
Correct Answer: (c)
In the other pairs, the first item is the monomer/building block of the second. Glycerol is a component of lipids, but not a monomer in the same way amino acids form proteins or nucleotides form DNA.
In the other pairs, the first item is the monomer/building block of the second. Glycerol is a component of lipids, but not a monomer in the same way amino acids form proteins or nucleotides form DNA.
Q134. When we homogenise any tissue in an acid the acid soluble pool represents
Correct Answer: (a)
The acid-soluble pool roughly represents the cytoplasmic composition, containing small molecules, while organelles and membranes are in the insoluble fraction.
The acid-soluble pool roughly represents the cytoplasmic composition, containing small molecules, while organelles and membranes are in the insoluble fraction.
Q135. When triglycerides are solid at ordinary room temperature, they are called
Correct Answer: (c)
Triglycerides that remain solid at room temperature are called fats (usually containing saturated fatty acids), while those that are liquid are called oils.
Triglycerides that remain solid at room temperature are called fats (usually containing saturated fatty acids), while those that are liquid are called oils.
Q136. In the composition of cellular mass, arrange the components- proteins(P), carbohydrates(C), lipids(L) and nucleic acids(N) in decreasing order of mass percentage.
Correct Answer: (b)
Average cellular composition: Proteins (10-15%) > Nucleic Acids (5-7%) > Carbohydrates (3%) > Lipids (2%).
Average cellular composition: Proteins (10-15%) > Nucleic Acids (5-7%) > Carbohydrates (3%) > Lipids (2%).
Q137. Many elements are found in living organisms either free or in the form of compounds. One of the following is not found in living organisms.
Correct Answer: (a)
While trace amounts exist in some specific organisms, silicon is negligible in the general biological composition of humans/animals compared to the crust.
While trace amounts exist in some specific organisms, silicon is negligible in the general biological composition of humans/animals compared to the crust.
Q138. For a protein to have a quaternary structure, it must
Correct Answer: (b)
Quaternary structure exists when a functional protein is composed of more than one polypeptide chain (subunits).
Quaternary structure exists when a functional protein is composed of more than one polypeptide chain (subunits).
Q139. The given diagrammatic representation shows one of the categories of small molecular weight organic compounds in the living tissues. Identify the category shown and one blank component marked as “X” in it.
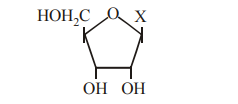

| Category | Component (X) |
|---|
Correct Answer: (d)
The diagram shows a ribose sugar with a nitrogenous base at 'X'. Since there is no phosphate group, it belongs to the category of nucleosides. Uracil is a base found in nucleosides of RNA.
The diagram shows a ribose sugar with a nitrogenous base at 'X'. Since there is no phosphate group, it belongs to the category of nucleosides. Uracil is a base found in nucleosides of RNA.
Q140. Natural lipids are readily soluble in
Correct Answer: (a)
Lipids are hydrophobic and dissolve readily in other non-polar substances like oils or organic solvents.
Lipids are hydrophobic and dissolve readily in other non-polar substances like oils or organic solvents.
Q141. Which one of the following statements is correct, with reference to enzymes?
Correct Answer: (a)
Holoenzyme (active) = Apoenzyme (protein) + Coenzyme/Cofactor (non-protein).
Holoenzyme (active) = Apoenzyme (protein) + Coenzyme/Cofactor (non-protein).
Q142. Choose the correct statement(s):
Correct Answer: (a)
Every metabolic reaction in a living system is catalyzed by enzymes; uncatalyzed reactions are too slow to sustain life. Note: biological rates often double/half for every 10°C change (Q10), not 37°C, and the living state is a non-equilibrium steady state.
Every metabolic reaction in a living system is catalyzed by enzymes; uncatalyzed reactions are too slow to sustain life. Note: biological rates often double/half for every 10°C change (Q10), not 37°C, and the living state is a non-equilibrium steady state.
Q143. Co-enzyme is
Correct Answer: (d)
Many co-enzymes are derived from vitamins (e.g., NAD and NADP contain niacin). They are non-protein organic cofactors.
Many co-enzymes are derived from vitamins (e.g., NAD and NADP contain niacin). They are non-protein organic cofactors.
Q144. Select the option which is not correct with respect to enzyme action:
Correct Answer: (b)
Competitive inhibition (like malonate vs succinate) is reversible; adding an excess of the substrate (succinate) will displace the inhibitor and restore activity.
Competitive inhibition (like malonate vs succinate) is reversible; adding an excess of the substrate (succinate) will displace the inhibitor and restore activity.
Q145. Choose the mismatched pair
Correct Answer: (c)
Valine is a non-polar, neutral amino acid, not a charged one.
Valine is a non-polar, neutral amino acid, not a charged one.
Q146. Consider the given statements:
(1) Amino acids can be classified as alpha, beta, gamma, delta based on functional group.
(2) Only L-amino acids are obtained on hydrolysis of proteins.
(3) Glycine shows high optical activity
Which of the statement(s) given above is/are correct?
(1) Amino acids can be classified as alpha, beta, gamma, delta based on functional group.
(2) Only L-amino acids are obtained on hydrolysis of proteins.
(3) Glycine shows high optical activity
Which of the statement(s) given above is/are correct?
Correct Answer: (d)
Proteins consist only of L-amino acids. Amino acids are classified based on the position of the amino group relative to the carboxyl group. Glycine is achiral and thus optically inactive.
Proteins consist only of L-amino acids. Amino acids are classified based on the position of the amino group relative to the carboxyl group. Glycine is achiral and thus optically inactive.
Q147. Which of the following is an antifertility steroid?
Correct Answer: (d)
Stilbesterol (Diethylstilbestrol) is a synthetic nonsteroidal estrogen often used in antifertility applications or hormone therapy.
Stilbesterol (Diethylstilbestrol) is a synthetic nonsteroidal estrogen often used in antifertility applications or hormone therapy.
Q148. Which of the following is not an attribute of enzymes ?
Correct Answer: (c)
Enzymes act as catalysts; they participate in the reaction but are released unchanged and can be used repeatedly.
Enzymes act as catalysts; they participate in the reaction but are released unchanged and can be used repeatedly.
Q149. The information in a genetic nucleic acid resides in the
Correct Answer: (c)
The genetic code is determined by the specific linear sequence of the four different nitrogenous bases in the nucleic acid chain.
The genetic code is determined by the specific linear sequence of the four different nitrogenous bases in the nucleic acid chain.
Q150. Carbohydrates, the most abundant biomolecule on earth, are produced by
Correct Answer: (a)
Carbohydrates are produced through photosynthesis by green plants, algae, and some photosynthetic bacteria.
Carbohydrates are produced through photosynthesis by green plants, algae, and some photosynthetic bacteria.
Q151. Consider the following statements:
(A) Coenzyme or metal ion that is tightly bound to enzyme protein is called prosthetic group.
(B) A complete catalytic active enzyme with its bound prosthetic group is called apoenzyme.
Select the correct option.
(A) Coenzyme or metal ion that is tightly bound to enzyme protein is called prosthetic group.
(B) A complete catalytic active enzyme with its bound prosthetic group is called apoenzyme.
Select the correct option.
Correct Answer: (b)
Statement (A) is correct. (B) is false because the active enzyme is the holoenzyme, not the apoenzyme.
Statement (A) is correct. (B) is false because the active enzyme is the holoenzyme, not the apoenzyme.
Q152. The Km value of the enzyme is the value of the substrate concentration at which the reaction reaches to
Correct Answer: (c)
The Michaelis constant (Km) is defined as the substrate concentration at which the reaction rate is exactly half of its maximum velocity (½ Vmax).
The Michaelis constant (Km) is defined as the substrate concentration at which the reaction rate is exactly half of its maximum velocity (½ Vmax).
Q153. Find out the odd one from the given pair.
Correct Answer: (d)
While DNA has hydrogen bonds between bases, the primary backbone of a polynucleotide is held together by phosphodiester bonds. The other pairs correctly identify the primary covalent linkage of the polymer.
While DNA has hydrogen bonds between bases, the primary backbone of a polynucleotide is held together by phosphodiester bonds. The other pairs correctly identify the primary covalent linkage of the polymer.
Q154. Linoleic acid is unsaturated fatty acid and its content is highest in
Correct Answer: (b)
Sunflower oil is known for its high content of polyunsaturated fatty acids, particularly linoleic acid.
Sunflower oil is known for its high content of polyunsaturated fatty acids, particularly linoleic acid.