Q1. COVID-19 is a single-stranded RNA virus. Which molecules would most likely be found in a single-stranded RNA virus, such as COVID-19?
Correct Answer: (d)
RNA contains ribose and uracil. Choice (A) is incorrect because deoxyribose and thymine are found in DNA, not RNA. Deoxyribose is found in DNA, not RNA, so choice (B) is incorrect. Choice (C) is incorrect because thymine is not found in RNA; it is found in DNA.
RNA contains ribose and uracil. Choice (A) is incorrect because deoxyribose and thymine are found in DNA, not RNA. Deoxyribose is found in DNA, not RNA, so choice (B) is incorrect. Choice (C) is incorrect because thymine is not found in RNA; it is found in DNA.
Q2. Catalase, an enzyme found in aerobic organisms, catalyzes the following reaction:
2H2O2 → 2H2O + O2
Predict what would happen if the catalase solution was boiled before performing the experiment, and justify your prediction.
2H2O2 → 2H2O + O2
Predict what would happen if the catalase solution was boiled before performing the experiment, and justify your prediction.
Correct Answer: (a)
Boiling denatures most enzymes, making them ineffective, and thus the reaction time would increase. Choice (B) is incorrect because some enzymes may be more effective at higher temperatures (for example, enzymes found in bacteria that live in warm environments). While increasing the temperature does increase the number of molecular collisions, choice (C) is incorrect because boiling would denature the enzyme, making it an ineffective catalyst. Choice (D) is incorrect because if the rate of the enzyme-catalyzed reaction was faster at higher temperatures, the average time for disks to float would decrease, not increase.
Boiling denatures most enzymes, making them ineffective, and thus the reaction time would increase. Choice (B) is incorrect because some enzymes may be more effective at higher temperatures (for example, enzymes found in bacteria that live in warm environments). While increasing the temperature does increase the number of molecular collisions, choice (C) is incorrect because boiling would denature the enzyme, making it an ineffective catalyst. Choice (D) is incorrect because if the rate of the enzyme-catalyzed reaction was faster at higher temperatures, the average time for disks to float would decrease, not increase.
Q3. BCR-ABL is an enzyme found in cancer cells in chronic myelogenous leukemia. ATP binds to the active site of BCR-ABL, which then stimulates cell division in cancer cells. Which of the following would most likely slow the rate of cell division in cancer cells with the BCR-ABL enzyme?
Correct Answer: (c)
Competitive inhibitors bind to the active site of an enzyme. Therefore, adding a competitive inhibitor of BCR-ABL would block ATP from binding to its active site. Choices (A) and (B) are both incorrect because cofactors and coenzymes enhance enzyme function. Choice (D) is incorrect because a transcription factor would not affect ATP’s ability to bind to the enzyme.
Competitive inhibitors bind to the active site of an enzyme. Therefore, adding a competitive inhibitor of BCR-ABL would block ATP from binding to its active site. Choices (A) and (B) are both incorrect because cofactors and coenzymes enhance enzyme function. Choice (D) is incorrect because a transcription factor would not affect ATP’s ability to bind to the enzyme.
Q4. Which of the following best describes the differences between saturated and unsaturated lipids?
Correct Answer: (c)
Saturated lipids have carbon atoms linked through single bonds and form straight chains; this makes saturated lipids easier to pack tightly and more likely to be solid at room temperature. Choice (A) is incorrect because saturated lipids do not have C=C double bonds. Choice (B) is incorrect because saturated lipids are not amphipathic (phospholipids are amphipathic). Choice (D) is incorrect because while unsaturated lipids do have C=C double bonds, saturated lipids do not. Also, both saturated and unsaturated lipids are hydrophobic.
Saturated lipids have carbon atoms linked through single bonds and form straight chains; this makes saturated lipids easier to pack tightly and more likely to be solid at room temperature. Choice (A) is incorrect because saturated lipids do not have C=C double bonds. Choice (B) is incorrect because saturated lipids are not amphipathic (phospholipids are amphipathic). Choice (D) is incorrect because while unsaturated lipids do have C=C double bonds, saturated lipids do not. Also, both saturated and unsaturated lipids are hydrophobic.
Q5. Which of the following statements about enzymes is true?
Correct Answer: (b)
Enzymes are made of protein, ribozymes are made of RNA, and both have a catalytic effect on chemical reactions. Choice (A) is incorrect because enzymes never affect whether a reaction is endergonic or exergonic. Not all enzymes have an optimum pH of 7, so choice (C) is incorrect. Choice (D) is incorrect because enzymes are never consumed in the reactions they catalyze.
Enzymes are made of protein, ribozymes are made of RNA, and both have a catalytic effect on chemical reactions. Choice (A) is incorrect because enzymes never affect whether a reaction is endergonic or exergonic. Not all enzymes have an optimum pH of 7, so choice (C) is incorrect. Choice (D) is incorrect because enzymes are never consumed in the reactions they catalyze.
Q6. Which of the following drives the formation of a protein’s secondary structure?
Correct Answer: (b)
Secondary structure is driven by the formation of hydrogen bonds between the carboxyl groups and amino groups in a polypeptide chain. Choice (A) is incorrect because hydrophobic interactions between R-groups influence tertiary and quaternary structures but not secondary structures. Choice (C) is incorrect because disulfide bridges are formed in tertiary and quaternary structures, not secondary structures. Choice (D) is incorrect because multiple subunits/domains are only present in quaternary structures.
Secondary structure is driven by the formation of hydrogen bonds between the carboxyl groups and amino groups in a polypeptide chain. Choice (A) is incorrect because hydrophobic interactions between R-groups influence tertiary and quaternary structures but not secondary structures. Choice (C) is incorrect because disulfide bridges are formed in tertiary and quaternary structures, not secondary structures. Choice (D) is incorrect because multiple subunits/domains are only present in quaternary structures.
Q7. In an aqueous environment like the cytosol, the most stable tertiary protein structures would have hydrophilic amino acids in which part of the protein’s structure?
Correct Answer: (c)
Tertiary protein structures in the cell are most stable when their hydrophilic amino acids are on the surface of the protein, in contact with the watery cytosol of the cell. Choices (A) and (B) are incorrect because hydrophobic amino acids are more likely to be found in the interior of a protein, away from water in the cytosol. Choice (D) is incorrect because hydrophilic amino acids would not avoid water; they would be more stable when interacting with water.
Tertiary protein structures in the cell are most stable when their hydrophilic amino acids are on the surface of the protein, in contact with the watery cytosol of the cell. Choices (A) and (B) are incorrect because hydrophobic amino acids are more likely to be found in the interior of a protein, away from water in the cytosol. Choice (D) is incorrect because hydrophilic amino acids would not avoid water; they would be more stable when interacting with water.
Q8. Which level of protein structure is formed by peptide bonds between amino acids?
Correct Answer: (a)
The primary structure of a protein is the sequence of amino acids held together by peptide bonds. Choice (B) is incorrect because secondary structure is formed by the hydrogen bonds between amino acids in a polypeptide chain. Tertiary structure is the globular shape formed by a polypeptide chain, so choice (C) is incorrect. Choice (D) is incorrect because quaternary structure is formed when multiple subunits come together to form the functional protein.
The primary structure of a protein is the sequence of amino acids held together by peptide bonds. Choice (B) is incorrect because secondary structure is formed by the hydrogen bonds between amino acids in a polypeptide chain. Tertiary structure is the globular shape formed by a polypeptide chain, so choice (C) is incorrect. Choice (D) is incorrect because quaternary structure is formed when multiple subunits come together to form the functional protein.
Q9. The molecular formula for glucose is C6H12O6. The molecule maltose is formed by a dehydration reaction that links two glucose molecules together. What is the molecular formula for maltose?
Correct Answer: (c)
A dehydration reaction removes a water molecule. Two glucose molecules would contain 12 carbon atoms, 24 hydrogen atoms, and 12 oxygen atoms, but the dehydration reaction (linking the two glucose molecules to form maltose) would remove two hydrogen atoms and one oxygen atom to form the water molecule removed in a dehydration reaction. This leaves 12 carbon atoms but only 22 hydrogen atoms and 11 oxygen atoms. Choice (A) is incorrect because it has fewer atoms than a single glucose molecule, so it could not be maltose (which is made from two glucose molecules). Choice (B) is incorrect because it is what would remain if a water molecule was removed from a single glucose molecule, not from two glucose molecules. Choice (D) is incorrect because it shows the number of atoms present in two glucose molecules without taking into account the atoms lost from the water molecule that was removed in the dehydration synthesis reaction.
A dehydration reaction removes a water molecule. Two glucose molecules would contain 12 carbon atoms, 24 hydrogen atoms, and 12 oxygen atoms, but the dehydration reaction (linking the two glucose molecules to form maltose) would remove two hydrogen atoms and one oxygen atom to form the water molecule removed in a dehydration reaction. This leaves 12 carbon atoms but only 22 hydrogen atoms and 11 oxygen atoms. Choice (A) is incorrect because it has fewer atoms than a single glucose molecule, so it could not be maltose (which is made from two glucose molecules). Choice (B) is incorrect because it is what would remain if a water molecule was removed from a single glucose molecule, not from two glucose molecules. Choice (D) is incorrect because it shows the number of atoms present in two glucose molecules without taking into account the atoms lost from the water molecule that was removed in the dehydration synthesis reaction.
Q10. Which of the following correctly describes the differences between competitive inhibitors and noncompetitive inhibitors?
Correct Answer: (a)
This statement accurately describes where competitive and noncompetitive inhibitors bind on enzymes. Choice (B) is incorrect because neither competitive inhibitors nor noncompetitive inhibitors bind to substrates or products. Choice (C) is incorrect because adding substrate can mitigate the effects of a competitive inhibitor, not a noncompetitive inhibitor. Both types of inhibitors reduce, not increase, the rate of enzyme-catalyzed reactions, so choice (D) is also incorrect.
This statement accurately describes where competitive and noncompetitive inhibitors bind on enzymes. Choice (B) is incorrect because neither competitive inhibitors nor noncompetitive inhibitors bind to substrates or products. Choice (C) is incorrect because adding substrate can mitigate the effects of a competitive inhibitor, not a noncompetitive inhibitor. Both types of inhibitors reduce, not increase, the rate of enzyme-catalyzed reactions, so choice (D) is also incorrect.
Q11. Which of the following correctly describes DNA but not RNA?
Correct Answer: (d)
Deoxyribose is found in DNA; ribose is found in RNA. Choice (A) is incorrect because DNA does not contain uracil. Choice (B) is incorrect because the hydroxyl group is attached to the 3′ carbon, not the 5′ carbon. Phosphodiester bonds are found in both DNA and RNA, so choice (C) is incorrect.
Deoxyribose is found in DNA; ribose is found in RNA. Choice (A) is incorrect because DNA does not contain uracil. Choice (B) is incorrect because the hydroxyl group is attached to the 3′ carbon, not the 5′ carbon. Phosphodiester bonds are found in both DNA and RNA, so choice (C) is incorrect.
Q12. Which of the following is common to both DNA and RNA?
Correct Answer: (c)
Both DNA and RNA have a phosphate group attached to the 5′ carbon. Choice (A) is incorrect because RNA does not contain thymine. A double-stranded antiparallel helix is only present in DNA, not RNA, so choice (B) is incorrect. Choice (D) is incorrect because ribose is present in RNA but not in DNA.
Both DNA and RNA have a phosphate group attached to the 5′ carbon. Choice (A) is incorrect because RNA does not contain thymine. A double-stranded antiparallel helix is only present in DNA, not RNA, so choice (B) is incorrect. Choice (D) is incorrect because ribose is present in RNA but not in DNA.
Q13. Which of the following best describes the formation of the primary structure of a protein?
Correct Answer: (b)
The primary structure of a protein is formed by linking amino acids, and dehydration reactions link amino acids in covalent bonds between the carboxyl group of one amino acid and the amino group of another amino acid. Choice (A) is incorrect because dehydration reactions do not form ionic bonds. Choices (C) and (D) are incorrect because hydrolysis reactions break, not form, bonds between monomers.
The primary structure of a protein is formed by linking amino acids, and dehydration reactions link amino acids in covalent bonds between the carboxyl group of one amino acid and the amino group of another amino acid. Choice (A) is incorrect because dehydration reactions do not form ionic bonds. Choices (C) and (D) are incorrect because hydrolysis reactions break, not form, bonds between monomers.
Q14. Refer to the following figure:
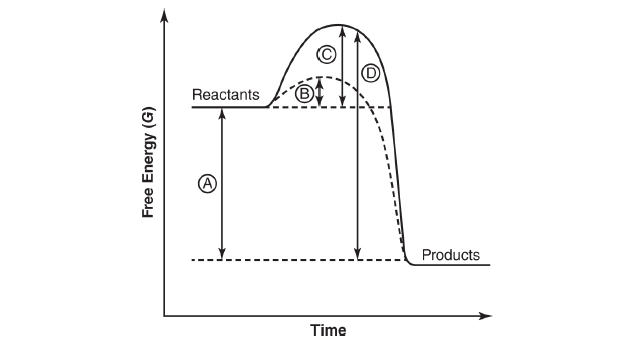
Which of the following represents the activation energy of an uncatalyzed reaction?

Which of the following represents the activation energy of an uncatalyzed reaction?
Correct Answer: (c)
The activation energy in the absence of an enzyme is the difference in free energy between the reactants and the transition state of the higher activation energy. Choice (A) is incorrect because it represents the overall free energy change of the reaction. Choice (B) is incorrect because it represents the activation energy of an enzyme-catalyzed reaction. Choice (D) represents the difference in free energy between the transition state of the reaction without the enzyme and the products of the reaction and is therefore incorrect.
The activation energy in the absence of an enzyme is the difference in free energy between the reactants and the transition state of the higher activation energy. Choice (A) is incorrect because it represents the overall free energy change of the reaction. Choice (B) is incorrect because it represents the activation energy of an enzyme-catalyzed reaction. Choice (D) represents the difference in free energy between the transition state of the reaction without the enzyme and the products of the reaction and is therefore incorrect.
Q15. Catalase, an enzyme found in aerobic organisms, catalyzes the following reaction:
2H2O2 → 2H2O + O2
A filter paper disk is saturated with the enzyme catalase and then placed at the bottom of a beaker of H2O2. As the reaction proceeds, oxygen bubbles will cling to the paper and eventually the paper will float to the top of the liquid. By measuring the time it takes for the catalase-saturated disk to float, one can compare the relative rates of decomposition of H2O2 under different experimental conditions.
An experiment was performed using catalase extracted from potatoes, with varying concentrations of H2O2. Multiple trials were conducted, and the means and the standard errors of the mean are shown in the following table:
Which of the following would be a suitable control for this experiment?
2H2O2 → 2H2O + O2
A filter paper disk is saturated with the enzyme catalase and then placed at the bottom of a beaker of H2O2. As the reaction proceeds, oxygen bubbles will cling to the paper and eventually the paper will float to the top of the liquid. By measuring the time it takes for the catalase-saturated disk to float, one can compare the relative rates of decomposition of H2O2 under different experimental conditions.
An experiment was performed using catalase extracted from potatoes, with varying concentrations of H2O2. Multiple trials were conducted, and the means and the standard errors of the mean are shown in the following table:
| Concentration of H2O2 | Mean Time for Disks to Float (seconds) | Standard Error of the Mean (seconds) |
|---|---|---|
| 1% | 92.6 | 6.2 |
| 3% | 32.5 | 5.9 |
| 6% | 15.1 | 4.5 |
Which of the following would be a suitable control for this experiment?
Correct Answer: (b)
Using a paper disk soaked in water would demonstrate the rate of reaction without the enzyme and would provide a basis for comparison. Choice (A) is incorrect because changing the temperature in just one of the beakers would introduce another variable into the experiment. Choice (C) is incorrect because this change would only lead to comparing the effectiveness of liver catalase versus that of potato catalase. Changing the pH in just one of the beakers would also introduce another variable into the experiment, so choice (D) is also incorrect.
Using a paper disk soaked in water would demonstrate the rate of reaction without the enzyme and would provide a basis for comparison. Choice (A) is incorrect because changing the temperature in just one of the beakers would introduce another variable into the experiment. Choice (C) is incorrect because this change would only lead to comparing the effectiveness of liver catalase versus that of potato catalase. Changing the pH in just one of the beakers would also introduce another variable into the experiment, so choice (D) is also incorrect.
Q16. A scientist conducted an experiment to find out what type of macromolecule a virus injects into a cell. Using radiolabeled atoms, the scientist found that phosphorus from the virus entered the cell but sulfur did not. Which of the following molecules would most likely be injected from this virus into the cell?
Correct Answer: (b)
Nucleic acids contain phosphorus but not sulfur. So if phosphorus entered the cell but sulfur did not, a nucleic acid was most likely injected. Choice (A) is incorrect because carbohydrates typically do not contain phosphorus. Proteins typically contain sulfur but not phosphorus, so choice (C) is incorrect. Choice (D) is incorrect because steroids are a type of lipid that do not contain phosphorus.
Nucleic acids contain phosphorus but not sulfur. So if phosphorus entered the cell but sulfur did not, a nucleic acid was most likely injected. Choice (A) is incorrect because carbohydrates typically do not contain phosphorus. Proteins typically contain sulfur but not phosphorus, so choice (C) is incorrect. Choice (D) is incorrect because steroids are a type of lipid that do not contain phosphorus.
Q17. Refer to the following figure:

Which of the following represents the overall free energy change in the reaction?

Which of the following represents the overall free energy change in the reaction?
Correct Answer: (a)
The overall free energy change of the reaction is the difference between the free energy of the products less the free energy of the reactants. Choice (B) is incorrect because it represents the activation energy of an enzyme-catalyzed reaction. Choice (C) is incorrect because it represents the activation energy of the reaction without the enzyme. Choice (D) represents the difference in free energy between the transition state of the reaction without the enzyme and the products of the reaction and is therefore not the right answer.
The overall free energy change of the reaction is the difference between the free energy of the products less the free energy of the reactants. Choice (B) is incorrect because it represents the activation energy of an enzyme-catalyzed reaction. Choice (C) is incorrect because it represents the activation energy of the reaction without the enzyme. Choice (D) represents the difference in free energy between the transition state of the reaction without the enzyme and the products of the reaction and is therefore not the right answer.
Q18. An experiment was performed using catalase extracted from potatoes, with varying concentrations of H2O2. Multiple trials were conducted, and the means and the standard errors of the mean are shown in the following table:
A student graphs this data, showing 95% confidence intervals. Based on the 95% confidence intervals, which concentrations of enzyme are least likely to have statistically significant differences?
| Concentration of H2O2 | Mean Time for Disks to Float (seconds) | Standard Error of the Mean (seconds) |
|---|---|---|
| 1% | 92.6 | 6.2 |
| 3% | 32.5 | 5.9 |
| 6% | 15.1 | 4.5 |
A student graphs this data, showing 95% confidence intervals. Based on the 95% confidence intervals, which concentrations of enzyme are least likely to have statistically significant differences?
Correct Answer: (c)
The upper limit of the 95% confidence interval for the 6% concentration of enzyme (15.1 + 2(4.5) = 24.1) is greater than the lower limit of the 95% confidence interval for the 3% concentration of enzyme (32.5 – 2(5.9) = 20.7), so their 95% confidence intervals overlap. When 95% confidence intervals overlap, it is not possible to say there is a statistically significant difference between the two groups. Thus, choice (C) is the correct answer. The 95% confidence intervals for the 1% and 3% enzyme concentrations do not overlap, so choice (A) is incorrect. Similarly, choice (B) is incorrect because the 95% confidence intervals for the 1% and 6% enzyme concentrations do not overlap. Choice (D) is incorrect because the 95% confidence intervals for the 1% and 3% and the 1% and 6% enzyme concentrations do not overlap and likely have statistically significant differences between them.
The upper limit of the 95% confidence interval for the 6% concentration of enzyme (15.1 + 2(4.5) = 24.1) is greater than the lower limit of the 95% confidence interval for the 3% concentration of enzyme (32.5 – 2(5.9) = 20.7), so their 95% confidence intervals overlap. When 95% confidence intervals overlap, it is not possible to say there is a statistically significant difference between the two groups. Thus, choice (C) is the correct answer. The 95% confidence intervals for the 1% and 3% enzyme concentrations do not overlap, so choice (A) is incorrect. Similarly, choice (B) is incorrect because the 95% confidence intervals for the 1% and 6% enzyme concentrations do not overlap. Choice (D) is incorrect because the 95% confidence intervals for the 1% and 3% and the 1% and 6% enzyme concentrations do not overlap and likely have statistically significant differences between them.
Q19. Which of the following correctly describes how enzymes increase the rate of a chemical reaction?
Correct Answer: (b)
Enzymes increase the rate of reactions by reducing the activation energy of the reaction. Choice (A) is incorrect because enzymes never affect the overall free energy change of the reaction. Choices (C) and (D) are incorrect because enzymes cannot change the free energy of the reactants, nor can they change the free energy of the products of a reaction.
Enzymes increase the rate of reactions by reducing the activation energy of the reaction. Choice (A) is incorrect because enzymes never affect the overall free energy change of the reaction. Choices (C) and (D) are incorrect because enzymes cannot change the free energy of the reactants, nor can they change the free energy of the products of a reaction.
Q20. Refer to the following figure:

Which of the following represents the activation energy of an enzyme-catalyzed reaction?

Which of the following represents the activation energy of an enzyme-catalyzed reaction?
Correct Answer: (b)
The activation energy in the presence of an enzyme is the difference in free energy between the reactants and the transition state in the presence of the enzyme. Choice (A) is incorrect because it represents the overall free energy change of the reaction. Choice (C) is incorrect because it represents the activation energy of the reaction without the enzyme. Choice (D) represents the difference in free energy between the transition state of the reaction without the enzyme and the products of the reaction and is therefore incorrect.
The activation energy in the presence of an enzyme is the difference in free energy between the reactants and the transition state in the presence of the enzyme. Choice (A) is incorrect because it represents the overall free energy change of the reaction. Choice (C) is incorrect because it represents the activation energy of the reaction without the enzyme. Choice (D) represents the difference in free energy between the transition state of the reaction without the enzyme and the products of the reaction and is therefore incorrect.